|
Sometimes a little bit of background knowledge can open up a new experimental approach. One that I have stumbled upon recently comes from a 2012 finding from the Dulac lab. As a side note, this was a very early paper in the lifetime of eLife and was the first major paper to put it on my radar. In this work, Santoro and Dulac provide data in support of a model in which repeated activation of olfactory receptors results in changed activity of a histone variant, eventually culminating in prolonged lifespan of these cells.
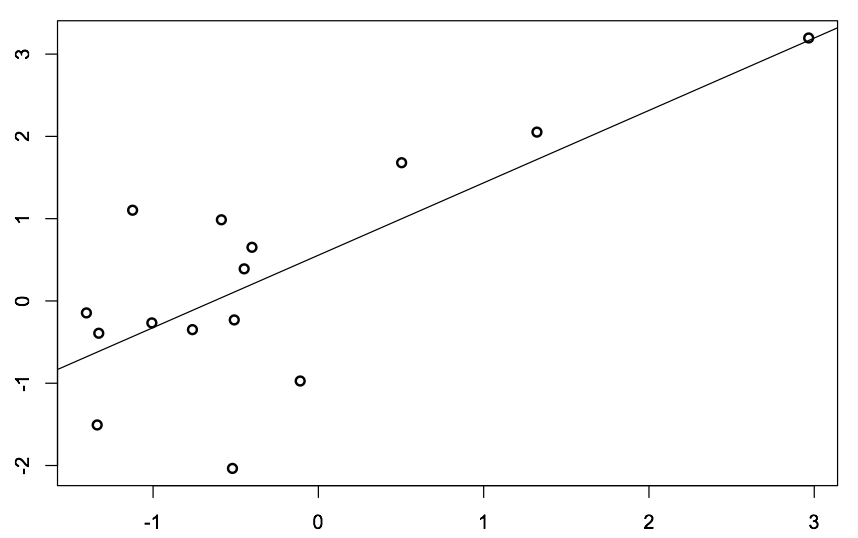
This is an interesting idea because it suggests a means by which an animal's receptor makeup could be modified by experience. The behavioral implications are not really as clear to estimate as one would like, given that increased representation of a given odor could either result in loss of sensitivity (via a background-type effect) or an increase in sensitivity, via an increased probability that an inhaled odorant will meet its receptor. An important follow-up on this work came last year from Ximena Ibarra-Soria and colleagues. Animals were chronically exposed to odors or had odors placed in their drinking water, such that they were only periodically and non persistently exposed. Surprisingly, in this work only the latter condition was sufficient to change the receptor repertoire. In other words, a background-type stimulus did not drive this change, while a more environmentally-relevant type of exposure did. The mechanistic details of these changes have yet to be enumerated but potentially offer a route by which to understand the complexity and dynamics of a tissue-level homeostat. Along the lines of these works. I analyzed some datasets from animals that had been repeatedly exposed to a stimulus, and to animals with a similar treatment, 5 weeks later. Below is a sample of those data, where I've hidden the axis titles, but where the y-axis corresponds to an activity value (>1 corresponding to activated), and the x-axis corresponds to the expression level of that gene 5 weeks later. As can be seen from the graph, efficiently enlisting olfactory receptors at the activity level correlates extremely tightly with overrepresentation of cells expressing that receptor at a later timepoint. For reference, animals were receiving a stimulus known to activate the three data points furthest to the right. To get to the original purpose of this post, this provides us with a potentially new and powerful experimental window. It may be sufficient to map ligands to receptors to simply expose animals as in the Ibarra-Soria study and then to measure receptor abundance later. Thus, an indirect approach at mapping ligand-receptor interactions based on the homeostatic biology of the tissue in question! |
 RSS Feed
RSS Feed